CH09 – Quantum Numbers

Dates
- P4 – Friday, September 15th
- P5 – Friday, September 15th
- P7 – Monday, September 18th
Orbitals – What do we know?
- where likely to be found
- “time-lapse” of electron position
- overlap of different electrons (because orbitals are not objects)
- could actually be anywhere
Orbitals – More definition
- within an atom, each electron has its own orbital “space”
- there are rules for where elctrons can be
- Characteristics: size, shape, orientation, spin
- Size: whole number ordinals, start with “1”, increase = more energy larger space
- Shape: s, p, d, f (indicate the shape of the orbital)
- Orientation: can be aligned with different axes
- Each orbital can hold 2 electrons, with different spin
- Each characteristic has a quantum muber (with rules)
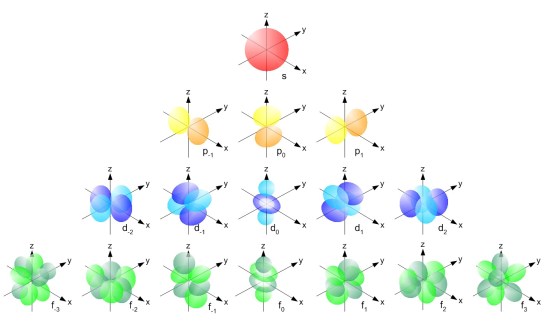
Coolest Web Site Ever
- The Orbitron [LINK]
- Can visualize the orbitals
- Also pictures in the book
How do write quantum mumbers
- QN Table
- Fill out table
- Write QN for lowest 25 electrons
Homework
- Finish QN table

You must be logged in to post a comment.