AP Chemistry – Boyle’s Law
One way to show a relationship between two variables is to find a way to plot a graph that provides a look at the data in linear fashion. In other words: how can I plot the data so that it gives a straight line? Straight lines are simply awesome, as they are easy to manipulate and extrapolate. They give us an easy way to look at what might be a complex relationship.
Consider the following data:
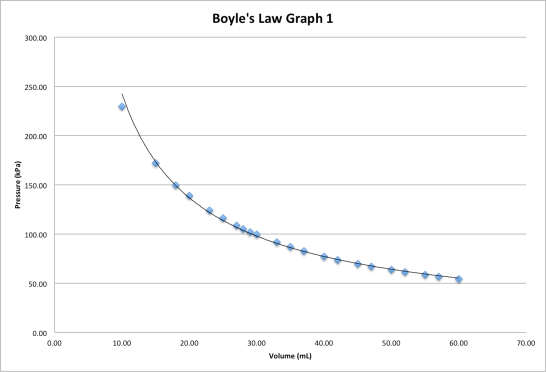
Notice that I have included two raw data columns as well as one calculated data column. This was done completely in MS Excel, easily the most powerful of the MS Office products. Also notice that the data seems out of order, as the volume numbers are out of sequence. This is not a problem as I plotted a “Scatter” plot using Excel. The result of a “P vs V” graph are shown below:
This is a classic P-V graph showing Boyle’s Law. And it does appear to give the inverse relationship which is characteristic of this law. However, how do we know how well we confirmed the law? Well, plotting the pressure as a function of (1/volume) is one way to do this and the results are shown below:
Now we get a cleaner look at the data. Notice that the line is, indeed, straight in the lower values. The data seems to go “off track” at high pressure situations. Does this make sense? Sure it does! The diligent scientist struggled to get that value and faced a tough challenge getting a data point at all. Otherwise, this data suggest that Boyle’s Law does apply to this gas, especially under low-pressure situations.



Pingback: CP48 – Boyle | Soxteacher