CH07 – Spectral Lines

Class Business
- a word about workload
- what to do when homework takes a long time
Textbook Notes
- goal – record big ideas
- student not expected to understand what is in chapter
- student is expected to know if an idea is present in chapter
- notes are another piece in understanding material
- final thought: if you are 45 minutes into HW and you are not “getting it” or have run out of time, please understand my homework policy, and how I would rather you sleep and come to school refreshed with questions.
Recap
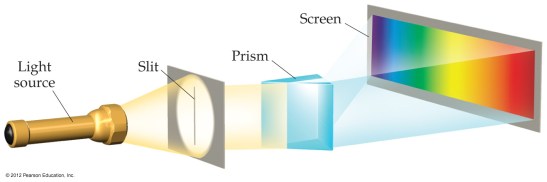
- the picture above is the result of a “continuous” energy spectrum
- continuous vs. quantized world
- can you think of any examples of quantized in your experience?
- it is difficult to process because we have no macro experience of this
- see above: Balmer (1885)
Energy Levels
- electrons can only have certain levels
- when given energy they rise accordingly
- not a stable scenario, with the electron returning to original level
- electrons will seek lowest energy possible
- will “step down”
- every possible combination of returning to lower level is seen
- when returning to lower levels, energy emitted as differences
- the energy emitted is LIGHT
- different energies of light = different colors
- LINES

Examples
- possible transitions with 4 levels
- what are emitted energies?
- what does energy spectrum look like?
Homework
- No HW
- We will use next class to determine spectrum
- please bring a calculator

You must be logged in to post a comment.